 Author
Author  Correspondence author
Correspondence author
International Journal of Molecular Ecology and Conservation, 2024, Vol. 14, No. 2
Received: 12 Feb., 2024 Accepted: 28 Mar., 2024 Published: 10 Apr., 2024
Reptiles are facing global ecological and genetic threats, leading many species to endangered status. Habitat loss, climate change, invasive species, pollution, and overexploitation are the primary ecological factors con-tributing to the decline of these species. Concurrently, genetic mechanisms such as genetic drift, reduced gene flow, genetic bottlenecks, and hybridization further exacerbate the extinction risks for these species. This study explores the ecological and genetic mechanisms driving reptile endangerment, analyzing how different ecological factors and genetic changes work together to lead to species decline and extinction. Through case studies, current conservation strategies and management approaches are discussed, and future research direc-tions are proposed. The study reveals that the intertwined ecological and genetic threats are collectively driv-ing the rapid decline of reptile species. The integration of landscape genetics and conservation genomics pro-vides new approaches and methods for the long-term conservation of these species. Understanding the eco-logical and genetic mechanisms of reptile endangerment is crucial for developing effective conservation measures. Combining ecological and genetic data analyses can help identify high-risk species and provide scientific evidence for formulating targeted conservation plans. Therefore, future research should focus on the application of emerging technologies, such as CRISPR and environmental DNA (eDNA), to enhance our un-derstanding of reptile genetic diversity and ecological adaptation.
1 Introduction
Reptiles play crucial roles in ecosystems, acting as both predators and prey, and contributing to various ecological processes such as gene dispersal, nutrient cycling, and ecosystem engineering. Their interactions within ecosystems are vital for maintaining ecological balance and biodiversity. For instance, large tropical reptiles significantly influence ecosystem functions through their trophic actions and other ecological interactions (Miranda, 2017). The loss of reptile species can lead to cascading effects on ecosystem health and stability, highlighting the importance of their conservation.
Reptiles are a diverse group of vertebrates, with approximately 10,000 species distributed across the globe. This diversity is unevenly spread, with tropical regions harboring the highest species richness (Brum et al., 2022). Despite their diversity, reptiles are often understudied compared to other vertebrate groups, leading to significant knowledge gaps in their ecology and conservation needs (Böhm et al., 2013; Brum et al., 2022). The uneven distribution of research effortsfurther complicates our understanding of reptile diversity and the specific threats they face in different regions (Brum et al., 2022).
The conservation status of reptiles is alarming, with nearly one in five species threatened with extinction. Factors such as habitat loss, climate change, pollution, and invasive species are major drivers of reptile declines. Additionally, many reptile species are classified as Data Deficient, indicating a lack of sufficient information to assess their conservation status accurately (Böhm et al., 2013). This data deficiency poses a significant challenge for conservation planning and action, necessitating innovative approaches to gather and analyze data on reptile populations and threats (Böhm et al., 2013; Nordstrom et al., 2022).
This study explores the ecological and genetic mechanisms that lead to the endangerment of reptiles. By synthesizing current research, it identifies key factors contributing to the decline in reptile populations and emphasizes effective conservation strategies. Understanding the ecological and genetic factors that contribute to reptile endangerment is crucial for developing effective conservation strategies. Through the discussion of various case studies, this study illustrates the challenges and successes in reptile conservation, providing insights into practical conservation strategies and their outcomes. Additionally, it highlights future research directions to address current knowledge gaps and improve reptile conservation efforts, enhancing the understanding and protection of reptile species globally.
2 Ecological Mechanisms of Endangerment
2.1 Habitat loss and fragmentation
Habitat loss and fragmentation are primary drivers of biodiversity decline globally. These processes reduce population sizes, limit gene flow, and increase the risk of inbreeding, which can lead to a loss of genetic diversity and increased extinction risk. Fragmentation often results from human activities such as deforestation, urbanization, and agricultural expansion, which create isolated habitat patches that are insufficient to support viable populations (Stow et al., 2001; Moore et al., 2008; Lino et al., 2019).
Deforestation, particularly in tropical regions, has led to significant habitat loss for many reptile species. For instance, the Amazonian lizards are facing severe habitat loss due to deforestation, which is compounded by climate change (Teixeira et al., 2022). Urbanization also poses a threat, as seen in the global assessment of urban ecology of reptiles, which highlights the uneven research distribution and the need for more studies to understand urban impacts on reptile conservation (Figure 1) (Brum et al., 2022). Agricultural expansion further fragments habitats, as evidenced by the reduced dispersal and genetic diversity in species like Cunningham’s skink in deforested areas (Stow et al., 2001).
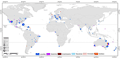 Figure 1 Proportional Distribution of Reptile Taxa in Global Urban Ecology Studies (Adapted from Brum et al., 2022) Image Caption: The figure illustrates the proportional distribution of reptile taxa involved in urban ecology studies across different global locations. Each pie chart represents a city or town, with the size of the pie chart indicating the number of studies conducted at that location. The different colored segments within the pie charts represent the various reptile taxa studied (e.g., lizards, snakes, turtles, crocodilians). Studies involving multiple taxa are represented by a "multiple taxa" category (Adapted from Brum et al., 2022) |
The study by Brum et al. (2022) examined the global distribution of reptile groups in urban ecology research, highlighting the focus and differences in research across various regions. The findings indicate that lizards hold a significant position in most urban ecology studies, while other reptiles, such as snakes and turtles, receive relatively less attention. Additionally, a few cities have research covering multiple reptile groups, reflecting the diversity and complexity of urban ecological studies in these areas. This global distribution map helps to understand the ecological roles of reptiles in different urban environments and provides a reference for future research.
2.2 Climate change
Climate change alters temperatures and weather patterns, which can disrupt the ecological balance and life cycles of reptiles. These changes can affect reproductive cycles, habitat suitability, and food availability, leading to population declines. Reptiles, being ectothermic, are particularly vulnerable to temperature fluctuations, which can impact their physiological processes and survival rates (Hu et al., 2020; Teixeira et al., 2022).
For example, Amazonian lizards are predicted to experience significant reductions in suitable habitats due to climate change, which will force them to shift their ranges or face extinction (Teixeira et al., 2022). Additionally, changes in temperature can alter reproductive cycles, as seen in squamate reptiles, where species with higher mean body temperatures and greater clutch sizes showed better post-disturbance responses (Hu et al., 2020).
2.3 Invasive species
Invasive species can outcompete native reptiles for resources, prey on them, or introduce diseases, leading to population declines. The introduction of non-native species often disrupts the existing ecological balance, making it difficult for native species to survive and reproduce (Hu et al., 2020; Brum et al., 2022).
Feral cats are known to prey on native reptiles, significantly impacting their populations. Invasive plants can alter habitat structures, making them unsuitable for native reptiles. For instance, the presence of invasive species in urban areas has been shown to affect the abundance and distribution of native reptile species, highlighting the need for targeted conservation efforts (Brum et al., 2022).
2.4 Pollution and contamination
Pollution and contamination are significant ecological mechanisms contributing to the endangerment of reptiles. Pesticides, heavy metals, and plastics are among the most pervasive pollutants affecting these species. Pesticides have been shown to cause a range of toxic effects in reptiles, including locomotor performance issues, histopathological changes, oxidative stress, neurotoxicology, and genetic damage (Freitas et al., 2020). Heavy metals and plastics, particularly endocrine-disrupting chemicals like bisphenol A (BPA), have been found to disrupt hormone signaling, leading to abnormalities in development, growth, and survival (Bhandari et al., 2015; Wu and Seebacher, 2020). These contaminants can cause sex steroid alterations, sex reversal, and developmental abnormalities, which are particularly concerning for species with temperature-dependent sex determination (Barraza et al., 2021).
Bioaccumulation and endocrine disruption are two critical examples of how pollution affects reptiles. Bioaccumulation occurs when contaminants build up in an organism over time, often leading to toxic effects. For instance, maternal transfer of contaminants has been observed across various reptile taxa, affecting reproductive success and offspring development (Barraza et al., 2021). Endocrine disruption, caused by chemicals like BPA and ethinyl estradiol (EE2), can lead to sex reversal and demasculinization of male-specific behaviors, which are essential for reproduction (Bhandari et al., 2015). These disruptions can have long-term effects on population dynamics and genetic fitness, potentially leading to population declines (Mingo et al., 2017).
2.5 Overexploitation
Overexploitation is another significant ecological mechanism of endangerment for reptiles. The collection of reptiles for the pet trade, traditional medicine, and food has led to substantial population declines. Many species are captured and sold in the pet trade, often leading to unsustainable harvesting rates. Additionally, reptiles are used in traditional medicine and as a food source in various cultures, further exacerbating their decline (Weir et al., 2010).
Specific examples of overexploitation include the harvesting of turtles and the skin trade in snakes. Turtles are particularly vulnerable due to their slow reproductive rates and long lifespans, making them susceptible to overharvesting. The demand for turtle meat and shells has led to significant population declines in many species. Similarly, the skin trade in snakes has resulted in the overexploitation of several species, with their skins being used for fashion and traditional medicine. This trade not only reduces population sizes but also disrupts local ecosystems by removing key predators and prey (Weir et al., 2010).
3 Genetic Mechanisms of Endangerment
3.1 Genetic drift and inbreeding
Small population sizes are particularly vulnerable to genetic drift, which can lead to a rapid loss of genetic diversity. This reduction in genetic diversity diminishes the population's ability to adapt to environmental changes and increases the likelihood of inbreeding. Inbreeding, in turn, can expose deleterious recessive alleles, leading to inbreeding depression and reduced fitness (Pavlova et al., 2017; Kyriazis et al., 2019; Pérez‐Tris et al., 2019).
Inbreeding depression has been observed in various reptile populations. For instance, in a fragmented population of the lizard Psammodromus algirus, increased homozygosity due to inbreeding was linked to reduced fitness, as evidenced by smaller adult body sizes and lower clutch masses (Pérez‐Tris et al., 2019). Similarly, the Macquarie perch, an endangered Australian freshwater fish, faces inbreeding depression due to small effective population sizes, which can be mitigated through assisted gene flow (Pavlova et al., 2017).
3.2 Reduced gene flow
Habitat fragmentation significantly impedes gene flow between populations, leading to genetic isolation. This isolation can result in reduced genetic diversity and increased genetic differentiation among populations. Fragmented habitats create physical barriers that prevent individuals from different populations from interbreeding, thereby limiting gene exchange (Honnay and Jacquemyn, 2007; Smith et al., 2009; Lino et al., 2019).
The endangered pygmy bluetongue lizard (Tiliqua adelaidensis) exhibits restricted gene flow due to habitat fragmentation, with significant genetic differentiation observed even between populations separated by short distances (Smith et al., 2009). Similarly, a meta-analysis of mammalian populations revealed that habitat fragmentation leads to decreased allelic diversity and heterozygosity, particularly in species with larger body mass and those dependent on forest habitats (Lino et al., 2019).
3.3 Bottlenecks and founder effects
Population bottlenecks occur when a population undergoes a drastic reduction in size, leading to a loss of genetic diversity. Founder effects arise when a new population is established by a small number of individuals from a larger population, resulting in reduced genetic variation in the new population. Both bottlenecks and founder effects can have long-term negative impacts on genetic diversity and population viability (Frankham, 2015; Lindsay et al., 2020).
An experimental study on sand lizards (Lacerta agilis) demonstrated that outbreeding in a newly founded island population eliminated malformations and improved hatching success compared to the inbred mainland population, highlighting the benefits of genetic diversity restoration (Lindsay et al., 2020). Additionally, a meta-analysis on genetic rescue showed that outcrossing inbred populations significantly increased fitness, particularly in stressful environments (Frankham, 2015).
3.4 Hybridization and introgression
Hybridization with non-native species can lead to genetic introgression, which may have both positive and negative effects on native populations. While hybrid vigor can enhance fitness, outbreeding depression may occur if the hybrids are less adapted to the local environment. The balance between these outcomes depends on the genetic compatibility and ecological context of the hybridizing species (Frankham, 2015; Lindsay et al., 2020).
In the case of the sand lizard, outbreeding with individuals from distant populations resulted in higher hatching success and elimination of malformations, demonstrating hybrid vigor (Lindsay et al., 2020). Conversely, the genetic rescue of small inbred populations often shows significant fitness benefits, but the risk of outbreeding depression must be carefully managed to avoid negative impacts (Frankham, 2015).
4 Integrating Ecological and Genetic Perspectives
4.1 Landscape genetics
Combining ecological data with genetic analysis is crucial for understanding how landscape features influence gene flow and genetic diversity in reptile populations. For instance, the study on the endangered pygmy bluetongue lizard (Tiliqua adelaidensis) demonstrated restricted gene flow across fragmented agricultural landscapes, highlighting the importance of habitat corridors to maintain genetic diversity and connectivity (Smith et al., 2009). Similarly, research on the jungle cat (Felis chaus) in central India used genome-wide SNP markers to reveal that roads and human-dominated land-use negatively impact gene flow, emphasizing the need for effective wildlife corridors (Tyagi et al., 2022). These examples underscore the value of integrating ecological and genetic data to inform conservation strategies.
4.2 Conservation genomics
The use of genomic tools in conservation planning has become increasingly important. Genomic analyses can identify adaptive genetic variation and inform strategies such as genomic rescue. For example, a landscape genomics study on the critically endangered Dahl's Toad-headed turtle (Mesoclemmys dahli) found that habitat fragmentation led to reduced gene flow and adaptive divergence, suggesting the need for genetic rescue to counteract genetic erosion and inbreeding (Gallego-García et al., 2019). Additionally, the genomes of two Extinct-in-the-Wild reptiles from Christmas Island provided insights into their evolutionary histories and highlighted the importance of maintaining genetic diversity in captive breeding programs to ensure successful reintroductions (Figure 2) (Dodge et al., 2023). These studies illustrate how conservation genomics can guide effective management and recovery efforts for endangered species.
.png) Figure 2 Overview of Genome Assemblies for Christmas Island Reptiles (Adapted from Dodge et al., 2023) Image Caption: (a) Displays the 18 longest contigs from the genome assembly of the blue-tailed skink, sorted by length, representing over 99% of the assembled sequence. (b) Shows the 22 longest scaffolds from the genome assembly of the Lister's gecko, sorted by length, representing over 96% of the assembled sequence. In each figure, red dots indicate the positions of chromosome telomeres, with dark gray regions representing contigs or scaffolds spanning from telomere to telomere. The dashed lines indicate the estimated chromosome numbers based on the karyotypes of closely related species. (Adapted from Dodge et al., 2023) |
Dodge et al. (2023) conducted a study that produced high-quality genome assemblies for two reptile species from Christmas Island using PacBio HiFi sequencing and Hi-C technology. The genome assemblies of the blue-tailed skink and Lister's gecko demonstrated high continuity and coverage, with some contigs and scaffolds extending from one telomere to the other. These genomic data are not only significant for understanding the genome structure and evolutionary relationships of these species but also provide valuable genomic resources for conservation biology.
4.3 Ecological niche modeling
Ecological niche modeling is a powerful tool for predicting habitat suitability and range shifts, particularly in the context of climate change. By modeling the impacts of climate change, researchers can identify potential refugia and prioritize areas for conservation. For instance, a review on the effects of habitat loss and fragmentation on amphibians emphasized the need for multi-scale, species-specific investigations to understand population connectivity and inform conservation strategies (Cushman, 2006). Moreover, the application of environmental DNA (eDNA) techniques has shown promise in detecting elusive species and assessing community diversity, which can enhance the accuracy of ecological niche models and improve conservation planning (Nordstrom et al., 2022). These approaches enable more precise predictions of habitat suitability and inform proactive measures to mitigate the impacts of environmental changes on reptile populations.
5 Case Analysis: The Plight of the Eastern Indigo Snake
5.1 Ecological challenges
The Eastern Indigo Snake (Drymarchon couperi) has experienced significant population declines primarily due to habitat loss, degradation, and fragmentation in the southeastern United States. Studies have shown that these snakes require large tracts of undeveloped land with a mix of xeric uplands and adjacent wetland habitats to thrive. Habitat fragmentation, particularly in suburban and highway-adjacent areas, has been linked to reduced survival rates, highlighting the critical need for conservation core areas to support their populations (Breininger et al., 2012; Hyslop et al., 2014).
Invasive species and climate change further exacerbate the ecological challenges faced by the Eastern Indigo Snake. Invasive species can alter the habitat structure and availability of prey, while climate change can affect the distribution and availability of suitable habitats. These factors combined with habitat fragmentation create a complex web of ecological pressures that threaten the survival of this species (Hyslop et al., 2014; Bauder et al., 2021).
5.2 Genetic challenges
The Eastern Indigo Snake populations are highly fragmented, leading to reduced genetic diversity and increased inbreeding. Genetic studies have revealed significant genetic differentiation among populations, with each population representing a unique genetic cluster. This isolation can result in small effective population sizes, which are susceptible to genetic drift and loss of adaptive variation (Krysko et al., 2016; Bauder et al., 2021).
Historical and contemporary population declines have led to genetic bottlenecks, further reducing genetic diversity. The genetic structure of these populations indicates that they have historically existed in small, isolated groups, which may have been exacerbated by recent anthropogenic impacts. This genetic bottleneck effect poses a significant challenge to the long-term viability of the species (Krysko et al., 2016; Sovic et al., 2019).
5.3 Conservation efforts
Conservation efforts for the Eastern Indigo Snake include habitat restoration and management to mitigate the effects of habitat loss and fragmentation. Protecting large tracts of undeveloped land and ensuring a mix of suitable habitats are crucial for the species' survival. Efforts are also focused on maintaining and restoring connectivity between fragmented habitats to facilitate gene flow and reduce the risk of inbreeding (Breininger et al., 2012; Hyslop et al., 2014; Bauder et al., 2021).
Genetic rescue and captive breeding programs are being implemented to address the genetic challenges faced by the Eastern Indigo Snake. These programs aim to increase genetic diversity and reduce inbreeding by introducing individuals from genetically diverse populations into isolated groups. Captive breeding programs also serve as a source for reintroduction efforts, helping to bolster wild populations (Krysko et al., 2016; Bauder et al., 2021).
5.4 Integrative strategies
Effective conservation of the Eastern Indigo Snake requires an integrative approach that combines ecological and genetic data. By understanding the species' habitat requirements and genetic structure, conservationists can develop targeted strategies to address both ecological and genetic challenges. This holistic approach ensures that conservation efforts are scientifically sound and effective in promoting the long-term survival of the species (Krysko et al., 2016; Bauder et al., 2021).
Ongoing conservation efforts have seen some successes, such as the establishment of protected areas and the implementation of translocation programs. However, challenges remain, including the need for continuous monitoring and adaptive management to respond to changing environmental conditions and emerging threats. The integration of ecological and genetic data into conservation planning is essential for overcoming these challenges and ensuring the persistence of the Eastern Indigo Snake (Breininger et al., 2012; Hyslop et al., 2014; Krysko et al., 2016; Bauder et al., 2021).
6 Conservation Strategies and Management
6.1 Habitat protection and restoration
The preservation of critical habitats is paramount for the conservation of endangered reptiles. Protecting and restoring these habitats ensures that reptiles have the necessary environment to thrive and maintain their populations. Establishing protected areas and habitat corridors are effective strategies to mitigate habitat fragmentation and promote gene flow among isolated populations. For instance, the study on the pygmy bluetongue lizard (Tiliqua adelaidensis) highlights the importance of habitat corridors to increase opportunities for gene flow and maintain genetic diversity in fragmented landscapes (Smith et al., 2009). Additionally, the creation of protected areas can safeguard essential habitats from further degradation and provide a refuge for endangered species.
6.2 Captive breeding and reintroduction
Captive breeding programs play a crucial role in maintaining genetic diversity and boosting populations of endangered reptiles. These programs provide a controlled environment where species can be bred and reared before being reintroduced into the wild. For example, the success of captive breeding and reintroduction efforts for the Christmas Island blue-tailed skink (Cryptoblepharus egeriae) and Lister's gecko (Lepidodactylus listeri) demonstrates the potential of these programs to expand populations from a few individuals to several thousand (Dodge et al., 2023). Head-start programs, where young reptiles are raised in captivity until they are less vulnerable to predation, have also shown success in increasing survival rates upon reintroduction. However, it is essential to manage the genetic adaptation to captivity to ensure that reintroduced individuals retain their fitness in natural environments (Williams and Hoffman, 2009).
6.3 Policy and legislation
Legal frameworks are vital in protecting reptiles and their habitats. Policies such as the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) and the Endangered Species Act (ESA) provide the necessary legal backing to regulate trade, protect critical habitats, and implement conservation measures. These frameworks help to prevent overexploitation and ensure that conservation efforts are legally supported. For instance, the ESA has been instrumental in the recovery of several reptile species by providing legal protection and funding for conservation programs (Shaffer et al., 2015).
6.4 Community engagement and education
Involving local communities in conservation efforts is crucial for the long-term success of reptile conservation. Community engagement and education can foster a sense of stewardship and encourage sustainable practices that benefit both the reptiles and the local people. Citizen science programs, where community members participate in monitoring and data collection, can provide valuable information for conservation management. Educational programs can raise awareness about the importance of reptiles and the threats they face, leading to increased support for conservation initiatives. For example, community-based conservation programs have been successful in promoting the protection of habitats and reducing human-wildlife conflicts (Towns et al., 2016).
7 Monitoring and Assessment Techniques
7.1 Population monitoring
Monitoring reptile populations is crucial for understanding their status and implementing effective conservation strategies. Various methods are employed to track population trends, each with its own advantages and limitations.
Mark-Recapture: This technique involves capturing individuals, marking them in a unique way, and then releasing them back into the wild. Subsequent recaptures allow researchers to estimate population size and survival rates. This method is particularly useful for species with distinct markings or those that can be safely tagged (Crane et al., 2022).
Telemetry: Biotelemetry involves attaching radio transmitters to individuals to track their movements and habitat use. This method compensates for several limitations in studying reptile natural history, such as their cryptic behavior and low detectability. Telemetry data can provide insights into home range sizes, movement patterns, and habitat preferences (Crane et al., 2022).
Camera Traps: Camera traps are passive monitoring devices that capture images or videos of animals as they pass by. This method is effective for detecting elusive or nocturnal species and can generate large datasets for analysis. Recent advancements in deep learning and computer vision have improved the accuracy of species identification from camera trap images, making this a valuable tool for monitoring reptile populations (Burton et al., 2015; Islam et al., 2023).
7.2 Genetic monitoring
Genetic monitoring techniques are essential for assessing the genetic diversity and structure of reptile populations, which are critical for their long-term survival and adaptability.
Microsatellite Analysis: Microsatellites are short, repetitive DNA sequences that are highly variable among individuals. Analyzing these markers can provide information on genetic diversity, population structure, and gene flow. This method is widely used in conservation genetics to identify distinct populations and assess their genetic health (Nordstrom et al., 2022).
Next-Generation Sequencing (NGS): NGS technologies allow for the rapid sequencing of entire genomes or specific genomic regions. This high-throughput approach can provide detailed insights into genetic variation, adaptive potential, and evolutionary history. NGS is particularly useful for studying non-model organisms and can be applied to both contemporary and historical DNA samples (Nordstrom et al., 2022).
7.3 Health and disease surveillance
Monitoring the health status and disease outbreaks in reptile populations is vital for preventing declines and managing conservation efforts.
Pathogen Screening: Regular screening for pathogens, such as viruses, bacteria, and parasites, can help detect and manage disease outbreaks. This involves collecting samples from individuals and analyzing them using molecular techniques to identify the presence of pathogens. Early detection of diseases can inform management actions to mitigate their spread (Nordstrom et al., 2022).
Health Assessments: Comprehensive health assessments involve evaluating the physical condition, nutritional status, and overall health of individuals. This can include physical examinations, blood tests, and other diagnostic procedures. Health assessments provide baseline data on the health of populations and can identify emerging health issues that may require intervention (Nordstrom et al., 2022).
8 Future Research Directions
8.1 Emerging technologies
The advent of new tools and techniques holds significant promise for the conservation of endangered reptiles. Technologies such as CRISPR and environmental DNA (eDNA) are at the forefront of this revolution. CRISPR, a powerful gene-editing tool, has the potential to correct genetic defects and enhance the resilience of endangered species to environmental stressors. Meanwhile, eDNA offers a non-invasive method to monitor species presence and biodiversity, which is particularly useful for elusive or cryptic species. For instance, eDNA has been successfully used to detect and monitor reptile populations, providing valuable data for conservation efforts (Thomsen and Willerslev, 2015; Adams et al., 2019; Nordstrom et al., 2022). As these technologies continue to advance, they are expected to become more sensitive, accessible, and integral to addressing key knowledge gaps in reptile conservation (Ruppert et al., 2019; Wang et al., 2020; Nordstrom et al., 2022).
8.2 Long-term and adaptive management
Continuous monitoring and flexible management strategies are crucial for the effective conservation of endangered reptiles. Adaptive management frameworks, which allow for iterative decision-making based on new data, are particularly valuable. Long-term ecological studies provide the necessary data to inform these frameworks, ensuring that conservation strategies remain relevant and effective over time. For example, the use of eDNA in long-term monitoring can help track changes in species distributions and population dynamics, providing critical insights for adaptive management (Barraza et al., 2021; Nordstrom et al., 2022; Yao et al., 2022). The integration of these approaches can help mitigate the impacts of environmental changes and anthropogenic pressures on reptile populations.
8.3 Interdisciplinary approaches
Integrating ecology, genetics, and social sciences is essential for holistic conservation planning. This interdisciplinary approach ensures that conservation strategies are comprehensive and consider ecological, genetic, and socio-economic factors. For instance, combining eDNA data with traditional ecological surveys and genetic analyses can provide a more complete picture of species distributions and population health (Thomsen and Willerslev, 2015; Farrell et al., 2022; Nordstrom et al., 2022). Additionally, involving stakeholders in conservation planning fosters collaboration and increases the likelihood of successful implementation. By bridging the gap between science and society, interdisciplinary approaches can enhance the effectiveness of conservation efforts and promote sustainable practices.
In summary, the future of reptile conservation lies in the integration of emerging technologies, long-term and adaptive management strategies, and interdisciplinary approaches. By leveraging these tools and frameworks, we can improve our understanding of the ecological and genetic mechanisms of endangerment and develop more effective conservation strategies to protect these vulnerable species.
9 Concluding Remarks
Reptile endangerment is driven by a combination of ecological and genetic mechanisms. Ecologically, habitat destruction, fragmentation, and environmental disturbances play significant roles in increasing extinction risks. These disturbances can lead to reduced population sizes, increased vulnerability to stochastic events, and disrupted ecological interactions. Genetically, small population sizes can result in inbreeding depression, loss of genetic variability, and fixation of deleterious mutations, further exacerbating extinction risks. Additionally, anthropogenic pollutants and endocrine disruptors have been shown to negatively impact reptile populations, highlighting the need for more targeted ecotoxicological studies.
Integrated conservation strategies that address both ecological and genetic factors are crucial. This includes habitat restoration, protection of critical habitats, and the establishment of new populations through translocations to predator-free islands. Moreover, understanding species-specific traits and their responses to environmental changes can help prioritize conservation efforts.
This study underscores the importance of a multifaceted approach to reptile conservation. By elucidating the ecological and genetic mechanisms of endangerment, it provides a comprehensive framework for developing effective conservation strategies. The findings have broader implications for biodiversity conservation, as reptiles play critical roles in ecosystem functioning, such as gene dispersal, nutrient cycling, and trophic interactions. Addressing the knowledge gaps in reptile ecotoxicology and the effects of environmental pollutants can also enhance the accuracy of ecological risk assessments and improve conservation outcomes.
Continued research and conservation efforts are essential to mitigate the risks facing reptile populations. This includes long-term ecological and genetic studies to monitor the success of conservation interventions and to adapt strategies as needed. Global cooperation and proactive measures are vital to address the widespread and multifaceted threats to reptiles. Conservationists, researchers, and policymakers must work together to implement effective conservation actions, raise public awareness, and secure the necessary resources for sustained efforts.
![]() Acknowledgments
Acknowledgments
Thanks to the peer reviewers for their suggestions on improving this study.
Conflict of Interest Disclosure
The authors affirm that this research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Adams C., Hoekstra L., Muell M., and Janzen F., 2019, A brief review of non-avian reptile environmental DNA (eDNA), with a case study of painted turtle (Chrysemys picta) eDNA under field conditions, Diversity. 11(4): 50.
https://doi.org/10.3390/d11040050
Barraza A., Finlayson K., Leusch F., and Merwe J., 2021, Systematic review of reptile reproductive toxicology to inform future research directions on endangered or threatened species, such as sea turtles, Environmental Pollution, 286: 117470 .
https://doi.org/10.1016/j.envpol.2021.117470
PMid:34438481
Bauder J., Peterman W., Spear S., Jenkins C., Whiteley A., and McGarigal K., 2021, Multiscale assessment of functional connectivity: Landscape genetics of eastern indigo snakes in an anthropogenically fragmented landscape in central Florida, Molecular Ecology, 30: 3422-3438.
https://doi.org/10.1111/mec.15979
PMid:33978288
Bhandari R., Deem S., Holliday D., Jandegian C., Kassotis C., Nagel S., Tillitt D., Saal F., and Rosenfeld C., 2015, Effects of the environmental estrogenic contaminants bisphenol A and 17α-ethinyl estradiol on sexual development and adult behaviors in aquatic wildlife species, General and comparative endocrinology, 214: 195-219.
https://doi.org/10.1016/j.ygcen.2014.09.014
PMid:25277515
Böhm M., Collen B., Baillie J., et al., 2013, The conservation status of the world's reptiles, Biological Conservation, 157: 372-385. Breininger D., Mazerolle M., Bolt M., Legare M., Drese J., and Hines J., 2012, Habitat fragmentation effects on annual survival of the federally protected eastern indigo snake, Animal Conservation, 15(4): 361-368.
https://doi.org/10.1111/j.1469-1795.2012.00524.x
Brum P., Gonçalves S., Strüssmann C., and Teixido A., 2022, A global assessment of research on urban ecology of reptiles: patterns, gaps and future directions, Animal Conservation, 26(1): 1-13.
https://doi.org/10.1111/acv.12799
Burton A., Neilson E., Moreira D., Ladle A., Steenweg R., Fisher J., Bayne E., and Boutin S., 2015, REVIEW: Wildlife camera trapping: a review and recommendations for linking surveys to ecological processes, Journal of Applied Ecology, 52: 675-685.
https://doi.org/10.1111/1365-2664.12432
Crane M., Silva I., Marshall B., and Strine C., 2020, Lots of movement, little progress: a review of reptile home range literature, Peer J, 9: e11742.
https://doi.org/10.7717/peerj.11742
PMid:34322323 PMCid:PMC8300531
Cushman S., 2006, Effects of habitat loss and fragmentation on amphibians: a review and prospectus, Biological Conservation, 128: 231-240.
https://doi.org/10.1016/j.biocon.2005.09.031
Dodge T., Farquharson K., Ford C., Cavanagh L., Schubert K., Schumer M., Belov K., and Hogg C., 2023, Genomes of two extinct-in-the-wild reptiles from christmas island reveal distinct evolutionary histories and conservation insights, Molecular Ecology Resources, 2023: 1-17.
https://doi.org/10.1111/1755-0998.13780
PMid:36872490
Farrell J., Whitmore L., Mashkour N., Ramia D., Thomas R., Eastman C., Burkhalter B., Yetsko K., Mott C., Wood L., Zirkelbach B., Meers L., Kleinsasser P., Stock S., Libert E., Herren R., Eastman S., Crowder W., Bovery C., Anderson D., Godfrey D., Condron N., and Duffy D., 2022, Detection and population genomics of sea turtle species via non-invasive environmental DNA analysis of nesting beach sand tracks and oceanic water, Molecular ecology resources, 22(7): 2471-2493.
https://doi.org/10.1111/1755-0998.13617
PMid:35377560
Frankham R., 2015, Genetic rescue of small inbred populations: meta-analysis reveals large and consistent benefits of gene flow, Molecular Ecology, 24(11): 2610-2618.
https://doi.org/10.1111/mec.13139
PMid:25740414
Freitas L., Paranaíba J., Pérez A., Machado M., and Lima F., 2020, Toxicity of pesticides in lizards, Human & Experimental Toxicology, 39: 596-604.
https://doi.org/10.1177/0960327119899980
PMid:31957493
Gallego-García N., Forero-Medina G., Vargas-Ramírez M., Caballero S., and Shaffer H., 2019, Landscape genomic signatures indicate reduced gene flow and forest‐associated adaptive divergence in an endangered neotropical turtle, Molecular Ecology, 28: 2757-2771.
https://doi.org/10.1111/mec.15112
PMid:31013379
Hyslop N., Meyers J., Cooper R., and Stevenson D., 2014, Effects of body size and sex of Drymarchon couperi (eastern indigo snake) on habitat use, movements, and home range size in Georgia, Journal of Wildlife Management, 78: 101-111.
https://doi.org/10.1002/jwmg.645
Honnay O., and Jacquemyn H., 2007, Susceptibility of common and rare plant species to the genetic consequences of habitat fragmentation, Conservation Biology, 21(3): 823-831.
https://doi.org/10.1111/j.1523-1739.2006.00646.x
PMid:17531059
Hu Y., Doherty T., and Jessop T., 2020, How influential are squamate reptile traits in explaining population responses to environmental disturbances? Wildlife Research, 47: 249-259.
https://doi.org/10.1071/WR19064
Islam S., Valles D., Hibbitts T., Ryberg W., Walkup D., and Forstner M., 2023, Animal species recognition with deep convolutional neural networks from ecological camera trap images, Animals: an Open Access Journal from MDPI, 13(9): 1526.
https://doi.org/10.3390/ani13091526
PMid:37174563 PMCid:PMC10177479
Krysko K., Nuñez L., Lippi C., Smith D., and Granatosky M., 2016, Pliocene-pleistocene lineage diversifications in the eastern indigo snake (Drymarchon couperi) in the Southeastern United States, Molecular Phylogenetics and Evolution, 98: 111-22.
https://doi.org/10.1016/j.ympev.2015.12.022
PMid:26778258
Kyriazis C., Wayne R., and Lohmueller K., 2019, Strongly deleterious mutations are a primary determinant of extinction risk due to inbreeding depression, Evolution Letters, 5: 33-47.
https://doi.org/10.1002/evl3.209
PMid:33552534 PMCid:PMC7857301
Lindsay W., Madsen T., Wapstra E., Lillie M., Loeb L., Ujvari B., and Olsson M., 2020, Long term effects of outbreeding: experimental founding of island population eliminates malformations and improves hatching success in sand lizards, Biological Conservation, 249: 108710.
https://doi.org/10.1016/j.biocon.2020.108710
Lino A., Fonseca C., Rojas D., Fischer E., and Pereira M., 2019, A meta-analysis of the effects of habitat loss and fragmentation on genetic diversity in mammals, Mammalian Biology, 94: 69-76.
https://doi.org/10.1016/j.mambio.2018.09.006
Mingo V., Lötters S., and Wagner N., 2017, The impact of land use intensity and associated pesticide applications on fitness and enzymatic activity in reptiles-A field study, The Science of the Total Environment, 590: 114-124.
https://doi.org/10.1016/j.scitotenv.2017.02.178
PMid:28259431
Miranda E., 2017, The plight of reptiles as ecological actors in the tropics, Frontiers in Ecology and Evolution, 5: 159.
https://doi.org/10.3389/fevo.2017.00159
Moore J., Miller H., Daugherty C., and Nelson N., 2008, Fine‐scale genetic structure of a long‐lived reptile reflects recent habitat modification, Molecular Ecology, 17(21): 4630-4641.
https://doi.org/10.1111/j.1365-294X.2008.03951.x
PMid:19140986
Nordstrom B., Mitchell N., Byrne M., and Jarman S., 2022, A review of applications of environmental DNA for reptile conservation and management, Ecology and Evolution, 12(6): e8995.
https://doi.org/10.1002/ece3.8995
PMid:35784065 PMCid:PMC9168342
Pavlova A., Beheregaray L., Coleman R., Gilligan D., Harrisson K., Ingram B., Kearns J., Lamb A., Lintermans M., Lyon J., Nguyen T., Sasaki M., Tonkin Z., Yen J., and Sunnucks P., 2017, Severe consequences of habitat fragmentation on genetic diversity of an endangered Australian freshwater fish: A call for assisted gene flow, Evolutionary Applications, 10: 531-550.
https://doi.org/10.1111/eva.12484
PMid:28616062 PMCid:PMC5469170
Pérez‐Tris J., Llanos-Garrido A., Bloor P., Carbonell R., Tellería J., Santos T., and Díaz J., 2019, Increased individual homozygosity is correlated with low fitness in a fragmented lizard population, Biological Journal of the Linnean Society, 128(4): 952-962.
https://doi.org/10.1093/biolinnean/blz144
Ruppert K., Kline R., and Rahman M., 2019, Past, present, and future perspectives of environmental DNA (eDNA) metabarcoding: A systematic review in methods, monitoring, and applications of global eDNA, Global Ecology and Conservation, 17: e00547.
https://doi.org/10.1016/j.gecco.2019.e00547
Shaffer H., Gidiş M., McCartney-Melstad E., Neal K., Oyamaguchi H., Tellez M., and Toffelmier E., 2015, Conservation genetics and genomics of amphibians and reptiles, Annual Review of Animal Biosciences, 3, 113-38.
https://doi.org/10.1146/annurev-animal-022114-110920
PMid:25580719
Smith A., Gardner M., Fenner A., and Bull C., 2009, Restricted gene flow in the endangered pygmy bluetongue lizard (Tiliqua adelaidensis) in a fragmented agricultural landscape, Wildlife Research, 36: 466-478.
https://doi.org/10.1071/WR08171
Sovic M., Fries A., Martin S., and Gibbs H., 2019, Genetic signatures of small effective population sizes and demographic declines in an endangered rattlesnake, Sistrurus catenatus, Evolutionary Applications, 12: 664 - 678.
https://doi.org/10.1111/eva.12731
PMid:30976301 PMCid:PMC6439488
Stow A., Sunnucks P., Briscoe D., and Gardner M., 2001, The impact of habitat fragmentation on dispersal of Cunningham's skink (Egernia cunninghami): evidence from allelic and genotypic analyses of microsatellites, Molecular Ecology, 10(4): 867-878.
https://doi.org/10.1046/j.1365-294X.2001.01253.x
PMid:11348496
Teixeira C., Trevelin L., Santos-Costa M., Prudente A., and Silva D., 2022, Synergistic effects of climate and landscape change on the conservation of Amazonian lizards, PeerJ, 10: e13028.
https://doi.org/10.7717/peerj.13028
PMid:35368330 PMCid:PMC8973465
Thomsen P., and Willerslev E., 2015, Environmental DNA-an emerging tool in conservation for monitoring past and present biodiversity, Biological Conservation, 183: 4-18.
https://doi.org/10.1016/j.biocon.2014.11.019
Towns D., Miller K., Nelson N., and Chapple D., 2016, Can translocations to islands reduce extinction risk for reptiles? Case studies from New Zealand, Biological Conservation, 204: 120-127.
https://doi.org/10.1016/j.biocon.2016.04.024
Tyagi A., Khan A., Thatte P., and Ramakrishnan U., 2022, Genome‐wide single nucleotide polymorphism (SNP) markers from fecal samples reveal anthropogenic impacts on connectivity: case of a small carnivore in the central Indian landscape. Animal Conservation, 25(5): 648-659.
https://doi.org/10.1111/acv.12770
Wang S., Yan Z., Hänfling B., Zheng X., Wang P., Fan J., and Li J., 2020, Methodology of fish eDNA and its applications in ecology and environment, The Science of the total environment, 755(2): 142622.、
https://doi.org/10.1016/j.scitotenv.2020.142622
PMid:33059148
Weir S., Suski J., and Salice C., 2010, Ecological risk of anthropogenic pollutants to reptiles: evaluating assumptions of sensitivity and exposure, Environmental pollution, 158(12): 3596-3606.
https://doi.org/10.1016/j.envpol.2010.08.011
PMid:20855139
Williams S., and Hoffman E., 2009, Minimizing genetic adaptation in captive breeding programs: a review, Biological Conservation, 142: 2388-2400.
https://doi.org/10.1016/j.biocon.2009.05.034
Wu N., and Seebacher F., 2020, Effect of the plastic pollutant bisphenol A on the biology of aquatic organisms: a meta‐analysis, Global Change Biology, 26: 3821-3833.
https://doi.org/10.1111/gcb.15127
PMid:32436328
Yao M., Zhang S., Lu Q., Chen X., Zhang S., Kong Y., and Zhao J., 2022, Fishing for fish environmental DNA: Ecological applications, methodological considerations, surveying designs, and ways forward, Molecular Ecology, 31: 5132-5164.
https://doi.org/10.1111/mec.16659
PMid:35972241

. HTML
Associated material
. Readers' comments
Other articles by authors
. Xinghao Li
. Jia Xuan
Related articles
. Large reptiles
. Endangerment mechanisms
. Ecological genetics
. Conservation genomics
. Ecological threats
Tools
. Post a comment


